The Structure and Chemistry of NMBs (UNDER CONSTRUCTION)
Neuromuscular blocking agents are able to bind to acetylcholine receptors and cause their specific effects because of the shape and structure of the molecules. In biology, the function of anything, from a small protein to a whole organ system, is determined by its structure. In chemistry, the type and arrangement of the different atoms making up a molecule determine its properties and how it will interact with other molecules. Neuromuscular blocking agent molecules and acetylcholine receptors are no exceptions to these rules. Here, you will learn how the arrangement of atoms in the neuromuscular blocking agent molecules allow them to bind to acetylcholine receptors.
The most important concept for you to know is that NMB molecules can bind to acetylcholine receptors in the same spot that acetylcholine would usually bind to because the NMBs have parts of their molecules that look like acetylcholine. If you know that much, you're usually good to go. But if you want to know more, read on!
This article assumes you understand that muscle contraction occurs from acetylcholine binding to acetylcholine receptors. If you need to know how acetylcholine and acetylcholine receptors work, click here. You should also know a bit of chemistry and biology. So without further ado...
The most important concept for you to know is that NMB molecules can bind to acetylcholine receptors in the same spot that acetylcholine would usually bind to because the NMBs have parts of their molecules that look like acetylcholine. If you know that much, you're usually good to go. But if you want to know more, read on!
This article assumes you understand that muscle contraction occurs from acetylcholine binding to acetylcholine receptors. If you need to know how acetylcholine and acetylcholine receptors work, click here. You should also know a bit of chemistry and biology. So without further ado...
Acetylcholine Structure
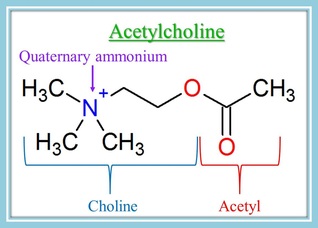
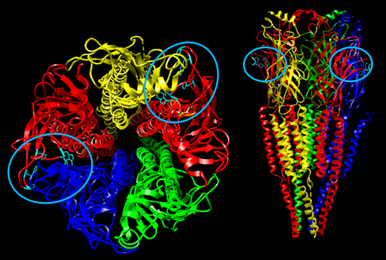
Acetylcholine receptors located on the muscles (known as nicotinic acetylcholine receptors) have two sites where acetylcholine binds to them. These areas are specially shaped so that the acetylcholine molecules fit into them.
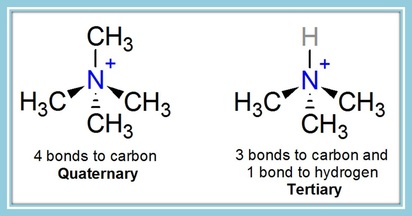
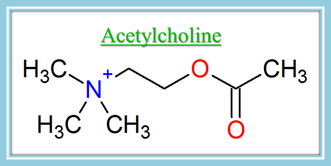
The acetylcholine molecule is named acetylcholine because it is made up of choline and an acetyl group. The most important part of this molecule for the purpose of comparing it to NMBs is the nitrogen you see on the left of the picture of acetylcholine. You may notice that this nitrogen has a positive charge on it. It also has 4 bonds to carbon atoms.
This positively charged nitrogen is the quaternary ammonium, and it causes a molecule to be highly attracted to the acetylcholine binding sites. It greatly helps both ACh and neuromuscular blockers bind to the nicotinic receptors.
What's the most important part of both acetylcholine and NMB molecules? Let me put it to you clearly.
N+ N+ N+ N+ N+ N+ N+ N+!!!!
This N+ nitrogen with 4 bonds to carbon is called a quaternary ammonium. It's named quaternary since there are 4 bonds to carbons, and ammonium meaning it's a nitrogen with the 4 bonds. To be a quaternary ammonium, all four bonds must be to an atom other than hydrogen.
The acetylcholine molecule is named acetylcholine because it is made up of choline and an acetyl group. The most important part of this molecule for the purpose of comparing it to NMBs is the nitrogen you see on the left of the picture of acetylcholine. You may notice that this nitrogen has a positive charge on it. It also has 4 bonds to carbon atoms.
This positively charged nitrogen is the quaternary ammonium, and it causes a molecule to be highly attracted to the acetylcholine binding sites. It greatly helps both ACh and neuromuscular blockers bind to the nicotinic receptors.
What's the most important part of both acetylcholine and NMB molecules? Let me put it to you clearly.
N+ N+ N+ N+ N+ N+ N+ N+!!!!
This N+ nitrogen with 4 bonds to carbon is called a quaternary ammonium. It's named quaternary since there are 4 bonds to carbons, and ammonium meaning it's a nitrogen with the 4 bonds. To be a quaternary ammonium, all four bonds must be to an atom other than hydrogen.
The Quaternary Ammonium
This positively charged nitrogen with 4 bonds to carbon is called a quaternary ammonium. It's named quaternary since there are 4 bonds to carbons, and ammonium meaning it's a nitrogen with the 4 bonds. To be a quaternary ammonium, all four bonds must be to an atom other than hydrogen.
Why is this N+ so important? Basically, it's the main part of the acetylcholine molecule that the receptor recognizes and depends on. The presence of a quaternary ammonium causes a molecule to be more highly attracted to acetylcholine binding sites. It greatly helps both acetylcholine and neuromuscular blockers bind to the nicotinic receptors.
All neuromuscular blocking molecules that are clinically used also have at least one quaternary ammonium so that they can effectively mimic that part of acetylcholine's structure and bind to the same spot on the acetylcholine receptors that acetylcholine normally would. Most also contain oxygen atoms a short distance away from the quaternary ammonium, which help them further mimic acetylcholine and bind to the receptors.
Since the N+ on neuromuscular blocking agents is positively charged, neuromuscular blocking agents are cations, positively charged ionic molecules that are very polar. Because of this, they can't pass through cell membranes and don't get absorbed easily into the bloodstream from the digestive system. Therefore, neuromuscular blocking agents must be injected to work. They also don't cross the blood brain barrier, which usually only lets small nonpolar molecules in, so they have no effect on consciousness. A solution containing a neuromuscular blocking agent will also contain a negative ion to balance the NMB's positive charge, usually chloride or bromide, and the clinincally used NMBs are all water soluble.
Neuromuscular blocking agents are often categorized by how many quaternary ammoniums (N+) they have.
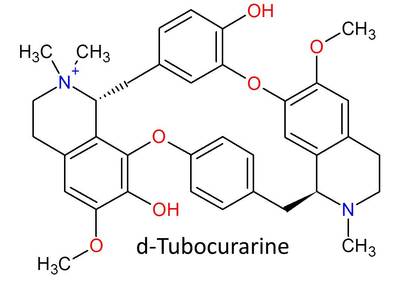
Note: An N+ with only 3 bonds to carbon and one bond to hydrogen is tertiary (meaning 3), not quaternary, even though it has total 4 bonds and a positive charge, because only 3 of its bonds are to carbons. I mainly mention this because sometimes d-tubocurarine is shown as having two positively charged nitrogens, but only one of those is quaternary with 4 bonds to carbons. All monoquaternary NMBs clinically used actually have two nitrogens - one quaternary, and one tertiary. The tertiary nitrogen only has 3 bonds to carbons. However, tertiary amines on neuromuscular blocking agent molecules are usually basic, so at physiological pH, they become protonated and gain a positive charge. This protonation further helps the molecules be attracted to bind to the receptors. In a more acidic environment, a greater the proportion of these molecules will be protonated, which is a reason why the effects of neuromuscular blocking agents are increased during acidosis.
Why is this N+ so important? Basically, it's the main part of the acetylcholine molecule that the receptor recognizes and depends on. The presence of a quaternary ammonium causes a molecule to be more highly attracted to acetylcholine binding sites. It greatly helps both acetylcholine and neuromuscular blockers bind to the nicotinic receptors.
All neuromuscular blocking molecules that are clinically used also have at least one quaternary ammonium so that they can effectively mimic that part of acetylcholine's structure and bind to the same spot on the acetylcholine receptors that acetylcholine normally would. Most also contain oxygen atoms a short distance away from the quaternary ammonium, which help them further mimic acetylcholine and bind to the receptors.
Since the N+ on neuromuscular blocking agents is positively charged, neuromuscular blocking agents are cations, positively charged ionic molecules that are very polar. Because of this, they can't pass through cell membranes and don't get absorbed easily into the bloodstream from the digestive system. Therefore, neuromuscular blocking agents must be injected to work. They also don't cross the blood brain barrier, which usually only lets small nonpolar molecules in, so they have no effect on consciousness. A solution containing a neuromuscular blocking agent will also contain a negative ion to balance the NMB's positive charge, usually chloride or bromide, and the clinincally used NMBs are all water soluble.
Neuromuscular blocking agents are often categorized by how many quaternary ammoniums (N+) they have.
- If a molecule has ONE quaternary ammonium, it is monoquaternary. d-Tubocurarine, rocuronium, and vecuronium are examples of monoquaternary NMBs.
- If it has TWO quaternary ammoniums, it's called bisquaternary. Pancuronium, atracurium, mivacurium, and doxacurium are examples of bisquaternary NMBs.
- If it has THREE quaternary ammoniums, it's triquaternary. The only triquaternary NMB that was ever used was gallamine, and it is no longer used today.
Note: An N+ with only 3 bonds to carbon and one bond to hydrogen is tertiary (meaning 3), not quaternary, even though it has total 4 bonds and a positive charge, because only 3 of its bonds are to carbons. I mainly mention this because sometimes d-tubocurarine is shown as having two positively charged nitrogens, but only one of those is quaternary with 4 bonds to carbons. All monoquaternary NMBs clinically used actually have two nitrogens - one quaternary, and one tertiary. The tertiary nitrogen only has 3 bonds to carbons. However, tertiary amines on neuromuscular blocking agent molecules are usually basic, so at physiological pH, they become protonated and gain a positive charge. This protonation further helps the molecules be attracted to bind to the receptors. In a more acidic environment, a greater the proportion of these molecules will be protonated, which is a reason why the effects of neuromuscular blocking agents are increased during acidosis.
Mimicking Acetylcholine
As I just mentioned but will repeat again (because this is important), neuromuscular blocking agents are mainly able to mimic acetylcholine and bind to acetylcholine receptors because they contain quaternary ammoniums. These quaternary ammoniums basically make them look just like acetylcholine. Most of the time, you can just look at a NMB molecule and tell what part of it looks like acetylcholine.
Let's try an easy one. Below is the structure of acetylcholine so you can use it for reference.
Let's try an easy one. Below is the structure of acetylcholine so you can use it for reference.
Here is a molecule of a neuromuscular blocking agent. You should easily be able to tell what parts of it look like ACh.
....
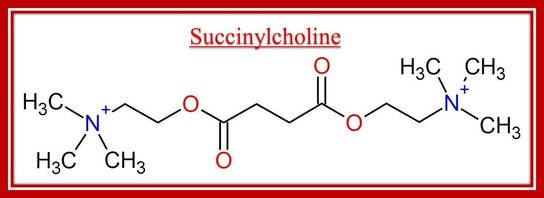
Well, that was easy, wasn't it? Succinylcholine is just two molecules of acetylcholine stuck together. It has two quaternary ammoniums, one on each side of the molecule. Because it looks so much like acetylcholine, it is able to bind to acetylcholine receptors in the same spot acetylcholine does. This specific NMB is a depolarizing one, meaning it actually activates the receptors and causes them to open and let ions through as if there really was acetylcholine in them. Other NMBs do not cause the acetylcholine receptor to open, and whether or not a NMB causes the acetylcholine receptor to open is also based on its structure. We'll get to structural differences between different neuromuscular blockers after an example of a non-depolarizing NMB.
Here is another neuromuscular blocking agent. This one is non-depolarizing. It binds to the acetylcholine receptors but does activate them or cause them to open. The picture on the right shows the parts of the molecule that mimic acetylcholine highlighted in blue.
Well, that was easy, wasn't it? Succinylcholine is just two molecules of acetylcholine stuck together. It has two quaternary ammoniums, one on each side of the molecule. Because it looks so much like acetylcholine, it is able to bind to acetylcholine receptors in the same spot acetylcholine does. This specific NMB is a depolarizing one, meaning it actually activates the receptors and causes them to open and let ions through as if there really was acetylcholine in them. Other NMBs do not cause the acetylcholine receptor to open, and whether or not a NMB causes the acetylcholine receptor to open is also based on its structure. We'll get to structural differences between different neuromuscular blockers after an example of a non-depolarizing NMB.
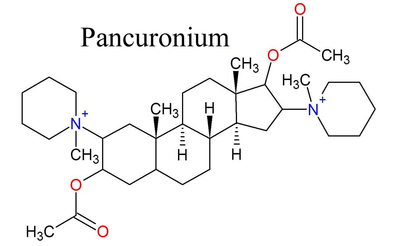
Here is another neuromuscular blocking agent. This one is non-depolarizing. It binds to the acetylcholine receptors but does activate them or cause them to open. The picture on the right shows the parts of the molecule that mimic acetylcholine highlighted in blue.
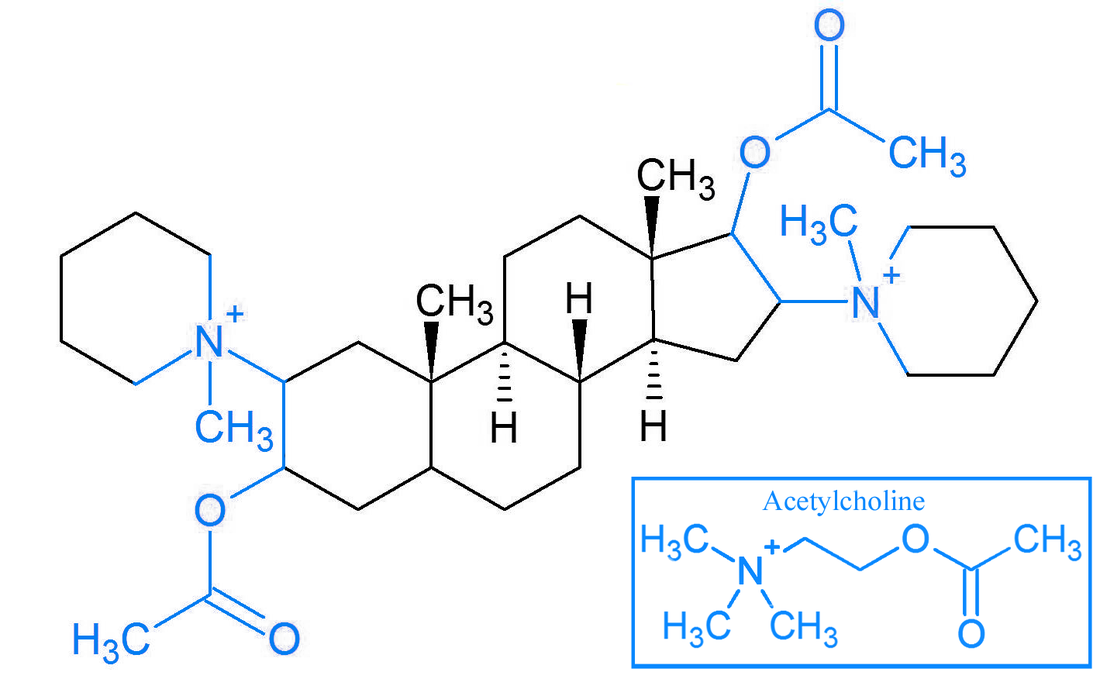
On the left and right sides of the molecule, you can once again see two quaternary ammoniums. Below and above the quaternary ammoniums, you can see acetyl groups (the things sticking out of the oxygens) that are also part of acetylcholine. Two molecules of acetylcholine were incorporated into this molecule, and the rigid steroid structure in the middle keeps them at a set distance from each other.
Pancuronium was originally designed for one molecule to bind to and block to both receptor sites at once, but it most likely actually just binds to one site at a time because the two binding sites in the actual acetylcholine receptor are now known to be further apart than the two acetylcholine-like parts of pancuronium. Either way though, pancuronium still works very well as a neuromuscular blocker.
Pancuronium was originally designed for one molecule to bind to and block to both receptor sites at once, but it most likely actually just binds to one site at a time because the two binding sites in the actual acetylcholine receptor are now known to be further apart than the two acetylcholine-like parts of pancuronium. Either way though, pancuronium still works very well as a neuromuscular blocker.
Depolarizing vs. Non-depolarizing structure
|
Now, why does a molecule like succinylcholine cause the receptor to open when it binds while other drugs like pancuronium don't? It's primarily because of the structure of the the molecules and the acetylcholine receptor.
The site on the receptor where acetylcholine binds to is a small pocket shaped to fit small, thin molecules like acetylcholine. If you've ever heard of the lock and key analogy for proteins binding to molecules, think of an acetylcholine receptor as having key holes, and acetylcholine is like the key that unlocks the receptor and causes it to open. Depolarizing agents are also thin molecules like acetylcholine, and the quaternary ammoniums may often simply contain methyl groups. Because of their slender shape, they fit into the binding site perfectly, the acetylcholine receptor is able to undergo a conformational change when they bind, and so the receptor opens just like it would for acetylcholine. In a way, these depolarizing agents are "keys" that are shaped in a way so that they also fit perfectly into the key hole of the ACh receptor and cause the receptor to open. |
Non-depolarizing NMBs, on the other hand, are close enough in structure to acetylcholine to bind to the receptors, but they do not fit exactly into the acetylcholine binding site, so they don't activate them like acetylcholine would. What they do instead is just prevent acetylcholine from binding to the receptors. A good analogy I've heard is that they are like glue that plugs up the key hole so you can't put the key in.
A demonstration!
If you want to better understand how some molecules can cause the acetylcholine receptor to open and change shape while other molecules can't, go get a small thin object like a pencil and a larger fat object like a ball (alternatively your other hand can work too.)
First, make a fist and then open the fist slightly so that your fingers and thumb form sort of a circle or hollow tube. The inside of your hand represents an acetylcholine binding site, which is like a little pocket within the protein channel. Get your pencil and place it into the hand Now, close your fist and wrap your fingers around it. Pretty easy, right? Normal acetylcholine is much like the pencil. When it binds to the binding site in the receptor, it causes the receptor to change shape, kind of like your hand changing shape as the fingers curl around the pencil. This change in the receptor shape when acetylcholine binds to it, called a conformational change, causes another part of the inside of the receptor that normally keeps the receptor closed, to twist open.
Depolarizing agents like succinylcholine are also simple, thin molecules without much random other bulk around their quaternary ammoniums. They're like the pencil - when a depolarizing agent binds to the acetylcholine receptor, the receptor still undergoes a conformational change because, for all it knows, that other depolarizing molecule is acetylcholine. Because of their thin shapes, depolarizing neuromuscular blocking agents are sometimes called leptocurares, the prefix lepto- meaning "thin".
Now, get the larger object or the fist of your other hand. Try to fit it into the little pocket your first hand made and then wrap your fingers around it. It's a bit more difficult, right? Non-depolarizing neuromuscular blocking agents work in a similar way. In a non-depolarizing NMB, the quaternary ammonium usually isn't just on the end with methyl groups (-CH3) attached to it; it's often part of some larger structure like a ring, and it has many other rings around it, giving it a more bulky shape. Therefore, it doesn't fit exactly into acetylcholine's binding site, and the acetylcholine receptor can't properly change shape to conform to the molecule with all that other stuff around it. So even though the molecule looks similar enough to acetylcholine that it is attracted to acetylcholine receptors, it isn't a perfect fit. Also, since non-depolarizing agents are just bigger and bulkier in general, they can also help physically block sodium ions from passing through the ACh receptor ion channel. Because of their bulky shape, non-depolarizing neuromuscular blocking agents are sometimes called pachycurares, the prefix pachy- meaning "thick".
Structures of Non-Depolarizing agents - steroid vs. BBTHIQ
While succinylcholine is the only depolarizing agent in use in the US today, there is a huge variety of non-depolarizing neuromuscular blocking agents that are currently used or have been used, and they are even further divided into two main groups based on their structure. Being in either of the two groups does NOT change the fact that these molecules are non-depolarizing or their mechanism of action. The names of the non-depolarizing agents vary slightly depending on their structure.
As you likely know, groups of drugs of the same type often have the same ending on their names. For example, local anesthetics usually end in "-caine" and beta blockers end usually in "-olol." Nondepolarizing NMBs usually have one of two drug name endings: "-curium" or "-curonium."
There are several NMBs that are their own unique structure, but the ones used today are one of two primary types of structures: aminosteroids (which end in "-curonium") and bis-benzyltetrahydroisoquinolines (which end in "-curium").
Steroid structure ("-curonium")
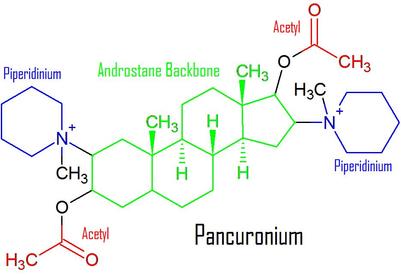
When I say "steroid," I'm talking of course about the shape of the molecule. Steroid molecules are typically made up of four carbon rings with different functional groups branching off of the rings. The steroid structured NMBs, however, are NOT actual lipid steroids like cholesterol or testosterone. They are something called aminosteroids, amino meaning they contain nitrogen. These NMBs have the same basic structural backbone, a steroid molecule called androstane. Pancuronium, the nondepolarizing molecule from above, has a steroid structure. All steroid-structured NMBs used today end in "-curonium." The main steroid structured NMBs that are used today are pancuronium, vecuronium, and rocuronium.
As you likely know, groups of drugs of the same type often have the same ending on their names. For example, local anesthetics usually end in "-caine" and beta blockers end usually in "-olol." Nondepolarizing NMBs usually have one of two drug name endings: "-curium" or "-curonium."
There are several NMBs that are their own unique structure, but the ones used today are one of two primary types of structures: aminosteroids (which end in "-curonium") and bis-benzyltetrahydroisoquinolines (which end in "-curium").
Steroid structure ("-curonium")
When I say "steroid," I'm talking of course about the shape of the molecule. Steroid molecules are typically made up of four carbon rings with different functional groups branching off of the rings. The steroid structured NMBs, however, are NOT actual lipid steroids like cholesterol or testosterone. They are something called aminosteroids, amino meaning they contain nitrogen. These NMBs have the same basic structural backbone, a steroid molecule called androstane. Pancuronium, the nondepolarizing molecule from above, has a steroid structure. All steroid-structured NMBs used today end in "-curonium." The main steroid structured NMBs that are used today are pancuronium, vecuronium, and rocuronium.
|
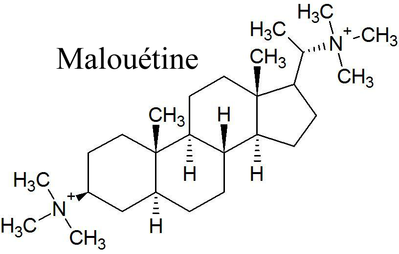
Steroid structured neuromuscular blocking agents were originally derived from a natural alkaloid called malouetine, from the plant Malouetia bequaertiana (good luck pronouncing that one). In the 1960's, scientists discovered that malouetine had neuromuscular blocking properties. And long story short, they found that it had a steroid structure separating its two quaternary ammoniums, and so they made several new derivatives with the rigid steroid structure to keep the quaternary ammoniums at a set distance apart. |
|
|
|
A typical steroid structured NMB today has one or two quaternary ammoniums branching off of its androstane backbone. These quaternary ammoniums are often part of a cyclic ring known in most cases. The first major clinically used steroid structured NMB like this was pancuronium. The name "pancuronium" actually stands for piperidino-androstane-curarizng-onium. (source)
To the right is the basic structure of pancuronium. Its quaternary ammoniums are incorporated into piperidinium rings. It also contains acetate esters, which help it minic acetylcholine. The end part of the ester excluding the single bonded oxygen will also be referred to as an acetyl group later on. |
Numbering the carbons in steroid NMBs
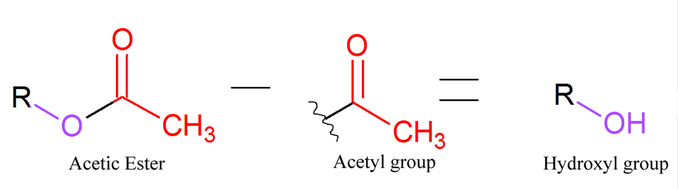
Whenever a patient is given a neuromuscular blocking agent, while some of the NMB molecules are binding to the receptors, others aren't binding to anything or they've recently unbound from a receptor, so they're just flowing around in the bloodstream. When a molecule is in your bloodstream, it will eventually be taken to the liver, where it may be metabolized in some way. Some steroid structured NMBs, namely the monoquaternary ones, are metabolized by the liver, where they can sometimes lose an acetyl group or two, depending on the molecule. Because of this, some metabolites are called des-acetyl (your NMB). "Des" is just another way to say "de". The acetyl group does not include the single bonded oxygen of the acetic ester, so when a molecule is de-acetylated, a hydroxyl group will be left behind.
This is shown in the simple equation below:
Whenever a patient is given a neuromuscular blocking agent, while some of the NMB molecules are binding to the receptors, others aren't binding to anything or they've recently unbound from a receptor, so they're just flowing around in the bloodstream. When a molecule is in your bloodstream, it will eventually be taken to the liver, where it may be metabolized in some way. Some steroid structured NMBs, namely the monoquaternary ones, are metabolized by the liver, where they can sometimes lose an acetyl group or two, depending on the molecule. Because of this, some metabolites are called des-acetyl (your NMB). "Des" is just another way to say "de". The acetyl group does not include the single bonded oxygen of the acetic ester, so when a molecule is de-acetylated, a hydroxyl group will be left behind.
This is shown in the simple equation below:
|
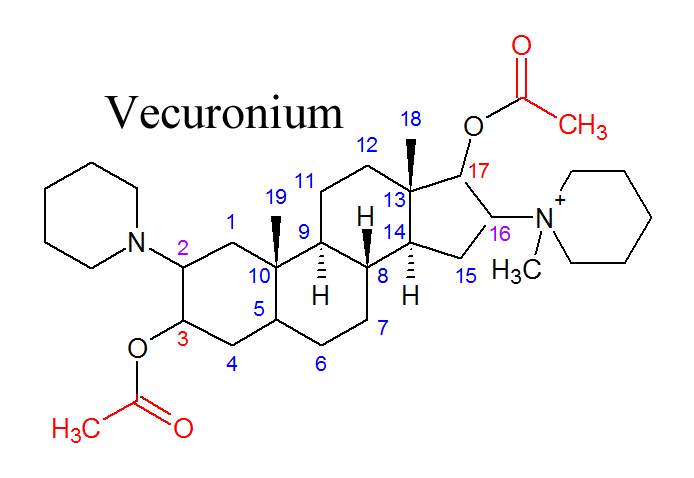
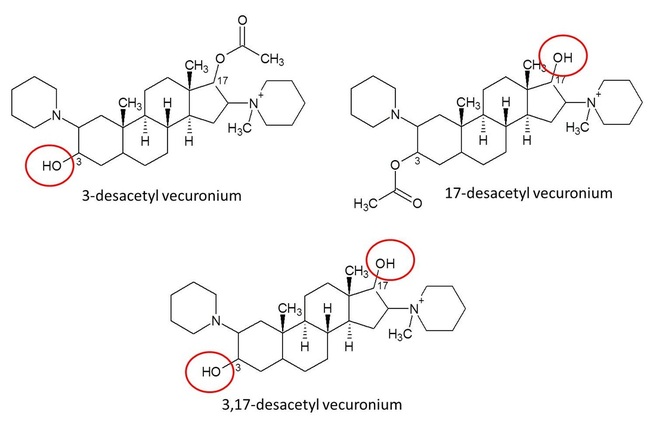
A steroid structured NMB has two possible places that an ester can go, carbon 3 or carbon 17. When a molecule is de-acetylated, it loses an acetyl group from the acetic ester. They can be coming off either carbon 3 or carbon 17 of the androstane molecule, so the metabolites are called 3-desacetyl or 17-desacetyl (your NMB). To the right is a picture of vecuronium, which is the best known NMB for losing acetyl groups when it is metabolized. Each of the carbons on vecuronium is labelled with its respective number, and the other steroid structured NMBs are labelled the same way. The acetyl group on the left of the picture coming off of the carbon labeled "3" is its 3-acetyl, and on the right coming off of "17" is its 17-acetyl. The carbons of the androstane skeleton are labeled, so you can see where carbons 3 and 17 are. |
When vecuronium loses an acetyl group, the new molecule is named after whatever acetyl group was lost.
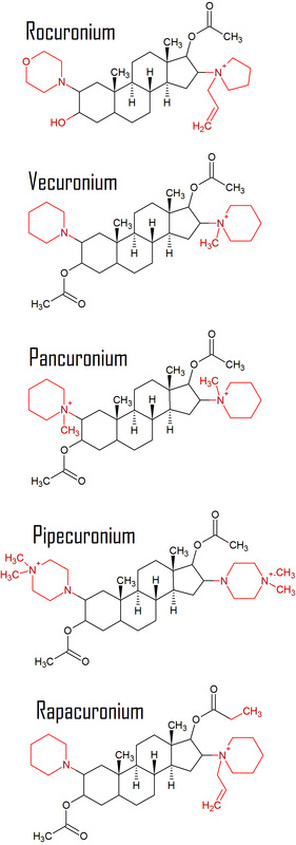
Examples of Steroid Structured NMBs (Distinguishing parts of the molecules are in red)
Below are all the steroid structured neuromuscular blocking agents that are or had previously been clinically used.
|
Rocuronium
|
|
Bis-benzyltetrahydroisoquinolinium structure (bbTHIQ)
Wow, that's a long name! What does it mean? Bis-benzyltetrahydroisoquinolines are some of the most beautiful molecules in existence. Sometimes, these molecules are just called benzylisoquinolines. This is wrong, but the term is most likely used since nobody wants to try to pronounce bis-benzyltetrahydroisoquinoline, and "benzylisoquinoline" is considered to be "close enough" to the correct term for people to understand what you're talking about. However, normal benzylisoquinolines are actually much less potent as NMBs than benzyltetrahydroisoquinolines. And now, you can now be one step ahead by knowing the meaning of the full correct name and in the process learn the structural details of these molecules.
Let's break it up:
Bis - benzyl - tetra hydro - iso quinoline
"Bis" is organic chemistry speak for "two." This will be important later on.
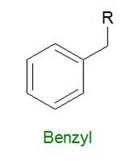
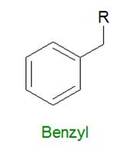
"Benzyl" refers to a benzyl functional group in organic chemistry. Benzyl looks like this:
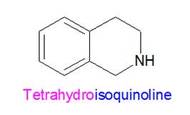
Tetrahydro and isoquinoline will be explained together.
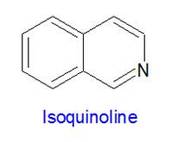
Isoquinoline is an organic molecule made up of two attached carbon rings, one of which contains a nitrogen atom.
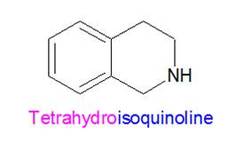
Tetrahydroisoquinoline is like isoquinoline except with 4 hydrogens (tetrahydro) bonded to some of the carbons and the nitrogen. As a result, those atoms now have more single bonds to hydrogens rather than double bonds to each other. These molecules looks like this:
Isoquinoline is an organic molecule made up of two attached carbon rings, one of which contains a nitrogen atom.
Tetrahydroisoquinoline is like isoquinoline except with 4 hydrogens (tetrahydro) bonded to some of the carbons and the nitrogen. As a result, those atoms now have more single bonds to hydrogens rather than double bonds to each other. These molecules looks like this:
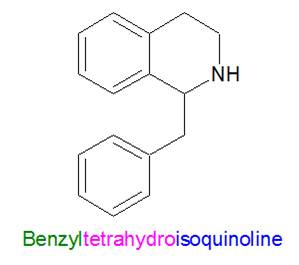
Now, if you add a benzyl group ..................... to a tetrahydroisoquinoline ........................... you get a benzyltetrahydroisoquinoline.
Finally, "bis" means that the molecule contains two benzyltetrahydroisoquinolines. The suffix "ium" often added to the end of the word indicates that the molecule is positively charged since it has the quaternary ammoniums, so the official full name of this type of molecule is bis-benzyltetrahydroisoquinolinium. Now, what does a bbTHIQ molecule look like?
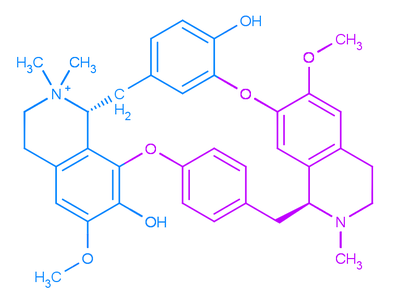
Below is d-tubocurarine, the very first and most historically important neuromuscular blocking agent, and it is considered to be a bis-benzyltetrahydroisoquinoline alkaloid. Take a good look at it. This molecule has both saved and ended lives, but since it was first introduced into medicine, it has saved many more than it has ever taken. In the righthand picture, I have highlighted the two benzyltetrahydroisoquinoline parts that come together to form the molecule.
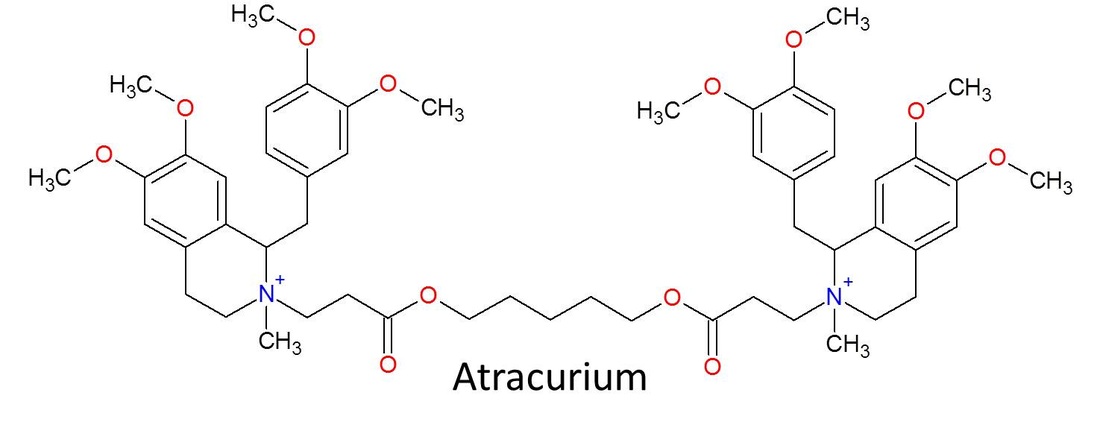
All other bis-benzyltetrohydroisoquinolinium derivatives used today have their structures derived from d-tubocurarine. They're the direct descendants of the original molecule of death, and that makes them pretty awesome. They also all end in "-curium." Some well-known bbTHIQ neuromuscular blockers are atracurium, cisatracurium, mivacurium, and doxacurium.
Atracurium and cisatracurium are still used regularly in the US today. However, all of these these new synthetic molecules have a much different structure than the original dTC.
Atracurium and cisatracurium are still used regularly in the US today. However, all of these these new synthetic molecules have a much different structure than the original dTC.
This is atracurium. It still has two benzyltetrahydroisoquinolines, but unlike with dTC, atracurium's are separate from each other and are instead connected by a nitrogen bridge, a chain with the two quaternary ammoniums on the ends. The bbTHIQ molecules are the largest of the NMB molecules, and they move and twist around a bit to be able to fit into the correct spots on the acetylcholine receptor. These molecules can also have several stereoisomers, basically different versions of the molecule where the atoms are basically in the same shape and order, but the 3D orientations of their atoms differ. Atracurium, for example, has 10 stereoisomers, and each of those isomers have varying degrees of potency and side effects. A solution of regular atracurium contains a mixture of each of those isomers. The picture above is not of any specific isomer, but it shows the overall general structure that the atracurium isomers follow.
Cisatracurium is a separate drug that is simply one of the 10 atracurium isomers, called the 1R-cis 1'R-cis isomer. The "cis" is referring to the bond to the benzyl group being in the same orientation as the bond going to the bridge. In atracurium, this cis-cis isomer makes up about 15% of the atracurium mixture. With cisatracurium, this one isomer exists by itself, rather than being part of the mixture of all 10. This cis-cis isomer by itself is about 4 times as potent as the overall atracurium drug mixture, and it also has fewer side effects. The only disadvantage cisatracurium has is that is has a slower onset and longer duration than atracurium, but overall, cisatracurium is considered to be safer and is used more often.
Cisatracurium is a separate drug that is simply one of the 10 atracurium isomers, called the 1R-cis 1'R-cis isomer. The "cis" is referring to the bond to the benzyl group being in the same orientation as the bond going to the bridge. In atracurium, this cis-cis isomer makes up about 15% of the atracurium mixture. With cisatracurium, this one isomer exists by itself, rather than being part of the mixture of all 10. This cis-cis isomer by itself is about 4 times as potent as the overall atracurium drug mixture, and it also has fewer side effects. The only disadvantage cisatracurium has is that is has a slower onset and longer duration than atracurium, but overall, cisatracurium is considered to be safer and is used more often.
Mivacurium is a bbTHIQ neuromuscular blocking agent that is no longer in clinical use. It has a short duration of about 15-20 minutes that would make it useful for shorter procedures, but it can also cause histamine release. Like the depolarizing drug succinylcholine, mivacurium is broken down by the enzyme pseudocholinesterase. Mivacurium as a drug is a mixture of 3 isomers, (E)-1R, 1'R ,2R, 2'R, cis-cis, (E)-1R, 1'R, 2R, 2'S, cis-trans, and (E)-1R, 1'R, 2S, 2'S, trans-trans, but the molecule itself can actually have 20 isomers due to the double bond in the center allowing for additional E/Z stereoisomerism. Unlike atracurium, mivacurium's cis-cis isomer by itself is actually the least potent one. Below is mivacurium, and you can cycle through its three main isomers by clicking the arrows in the upper righthand corner of the picture.
For stereochemistry names such as 1R, 2R, 1'R, 2'R, the 1 refers to the carbon adjacent to the nitrogen that the benzyl group is attached to. The 2 refers to the nitrogen itself. 1' and 2' refer to the carbon and nitrogen on the other side on the molecule. In the pictures, 1 is on the left side, and 1' is on the right side of the molecule.
Doxacurium has a molecular structure similar to mivacurium, and it is the longest lasting and most potent of the neuromuscular blockers that have been clinically used. It could last for a few hours, but it is no longer used in the US today, most likely due to its very long duration. Its release may also have been badly timed since mivacurium, which had a shorter duration, was FDA approved just one year after doxacurium. On the plus side, it has very few side effects. Doxacurium, like mivacurium, is used as a mixture of three isomers, but for doxacurium, they are all trans-trans isomers. Below is doxacurium, and you can cycle through its three main isomers by clicking the arrows in the upper righthand corner of the picture.
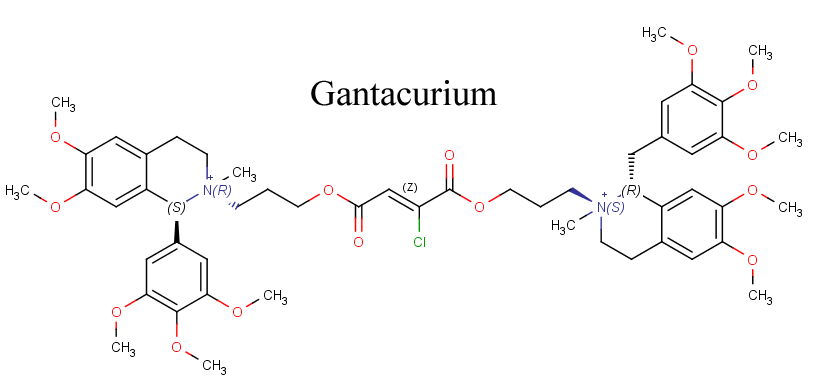
Gantacurium
One of the newer neuromuscular blocking agents that is still in development is a molecule called gantacurium. Although it is very similar in appearance to the other bbTHIQ drugs, it technically is not one. If you look on the left of the drawing of this molecule below, it does not have a benzyl group on one of its tetrahydroisoquinolines, but rather a phenyl group. Instead, gantacurium is part of a new in development group of drugs called chlorofumarates. Similarly to cisatracurium, gantacurium is only composed of one of its isomers, a trans-trans isomer.
One of the newer neuromuscular blocking agents that is still in development is a molecule called gantacurium. Although it is very similar in appearance to the other bbTHIQ drugs, it technically is not one. If you look on the left of the drawing of this molecule below, it does not have a benzyl group on one of its tetrahydroisoquinolines, but rather a phenyl group. Instead, gantacurium is part of a new in development group of drugs called chlorofumarates. Similarly to cisatracurium, gantacurium is only composed of one of its isomers, a trans-trans isomer.
Other structures of NMBs - Section under construction
Neuromuscular blocking agents have many very interesting structures. Although the ones that are clinically used today are either steroid or bis-benzyltetrahydroisoquinolinium structures (or succinylcholine), there are some other rather unique NMBs that have also existed.
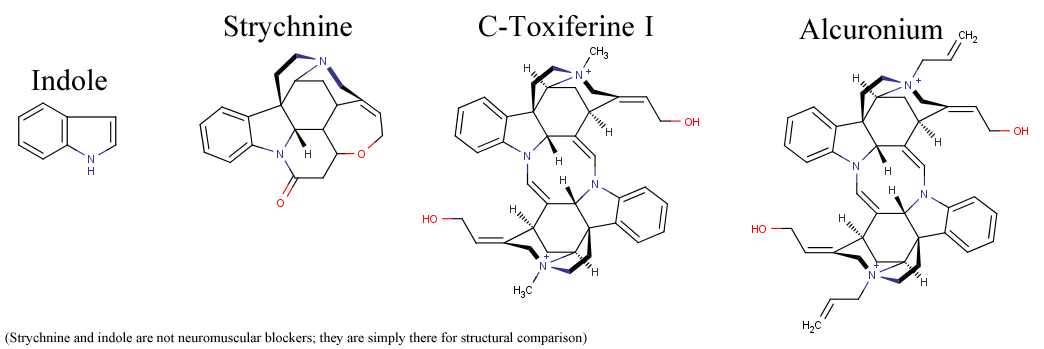
C-Toxiferine I and Alcuronium
Below is a molecule known as C-Toxiferine I, which is often just called toxiferine (in reality, there are over 30 toxiferine-like alkaloids, and the alkaloids named toxiferine go all the way up to Toxiferine XII, but C-Toxiferine I is the most well studied). Like d-tubocurarine, it is a curare alkaloid and is found in nature. It has a bis-indole structure. It comes from the the plant Strychnos toxifera, and yes, that's the same Strychnos that produces strychnine. In fact, the structure of toxiferine shows many similarities to strychnine. In addition, strychnine itself binds to glycine receptors, which are in the same protein family (cys loop receptors) and are very similar in structure to nicotinic acetylcholine receptors.
However, toxiferine, being a neuromuscular blocker, has pretty much the opposite effect as strychnine. It is also much more potent and has less side effects than d-tubocurarine. It most likely isn't as well-known as dTC though because it was isolated after dTC was, and it had a longer duration. Even after toxiferine was isolated, it's likely that people just chose to stick with dTC since it was good enough at the time and they were already familiar with it, so toxiferine never really saw clinical use. It had one derivative, alcuronium, which is less potent and more well known, but alcuronium was never actually clinically used in the US either. Alcuronium has the same structure as toxiferine, except it has allyl groups added to its quaternary ammoniums, hence the name AL-curonium.
C-Toxiferine I and Alcuronium
Below is a molecule known as C-Toxiferine I, which is often just called toxiferine (in reality, there are over 30 toxiferine-like alkaloids, and the alkaloids named toxiferine go all the way up to Toxiferine XII, but C-Toxiferine I is the most well studied). Like d-tubocurarine, it is a curare alkaloid and is found in nature. It has a bis-indole structure. It comes from the the plant Strychnos toxifera, and yes, that's the same Strychnos that produces strychnine. In fact, the structure of toxiferine shows many similarities to strychnine. In addition, strychnine itself binds to glycine receptors, which are in the same protein family (cys loop receptors) and are very similar in structure to nicotinic acetylcholine receptors.
However, toxiferine, being a neuromuscular blocker, has pretty much the opposite effect as strychnine. It is also much more potent and has less side effects than d-tubocurarine. It most likely isn't as well-known as dTC though because it was isolated after dTC was, and it had a longer duration. Even after toxiferine was isolated, it's likely that people just chose to stick with dTC since it was good enough at the time and they were already familiar with it, so toxiferine never really saw clinical use. It had one derivative, alcuronium, which is less potent and more well known, but alcuronium was never actually clinically used in the US either. Alcuronium has the same structure as toxiferine, except it has allyl groups added to its quaternary ammoniums, hence the name AL-curonium.
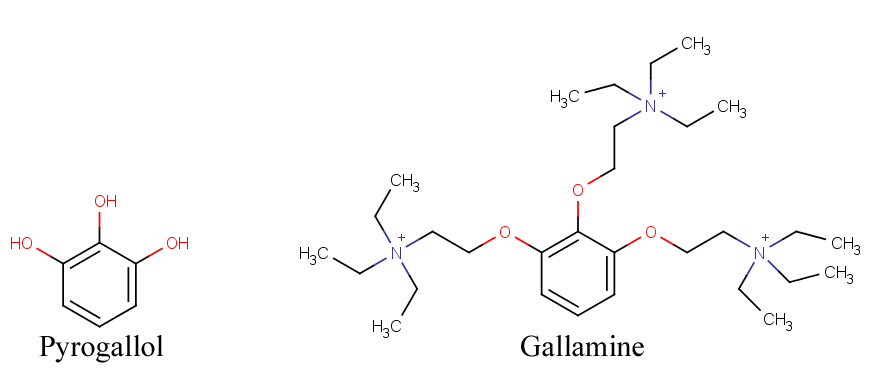
Gallamine
Gallamine was the very first synthetic neuromuscular blocker, developed in 1947 by Daniel Bovet, who later won a Nobel Prize for his work. It is derived from pyrogallol, a benzene ring with three adjacent hydroxyl groups, hence the name GALLamine. It is also the only tri-quaternary neuromuscular blocker. Its full name is "gallamine triethiodide" because gallamine alone can also refer to the tertiary amine version of the molecule where each nitrogen only has two ethyl groups on it instead of three. However, if it reacts with ethyl iodide, it forms the quaternary ammonium structure you know and love. Unfortunately, gallamine had significant vagolytic effects, meaning it increased the heart rate, and it released histamine at high doses. It also crosses the placenta, and its duration is greatly increased in renal failure. For these reasons, it was replaced by safer drugs.
A side note: There's a common misconception that N-N distance (the distance between the two nitrogens or quaternary ammoniums on a molecule) has a huge impact on potency for neuromuscular blockers, namely, that a further N-N distance makes a molecule more potent. N-N distance is indeed very important in depolarizing agents, where 10 carbons between nitrogens has been found to be the best distance for more potent molecules. For non-depolarizing neuromuscular blockers, however, the overall structure of the molecule itself is just as important, if not more important than the N-N distance. For example:
- The most potent curare alkaloid known is C-Alkaloid E, a molecule similar in structure to toxiferine. It is at least 100 times as potent as d-tubocurarine in terms of neuromuscular blocking activity. It has a N-N distance of about 7.78 angstroms. d-Tubocurarine, on the other hand, has a N-N distance of about 9.55 Angstroms, which is greater than that of C-Alkaloid E. However, despite the greater N-N distance, dTC is not more potent. (Distance values for both molecules were calculated in MarvinSketch).
- N-N distance also cannot fully account for potency differences in stereoisomers. d-Tubocurarine and l-tubocurarine have the same N-N distance. Yet d-tubocurarine is about 20 times more potent than l-tubocurarine.
- The two molecules toxiferine and alcuronium are almost the same molecule, except that alcuronium has an additional allyl group on its quaternary ammoniums where toxiferine has methyl groups. Alcuronium is much less potent than toxiferine. Both have the same N-N distance. In fact, rocuronium and rapacuronium are some of the least potent of the NMBs, and they have allyl groups as well. The addition of a simple functional group seems to have more impact than the N-N distance.
All molecular structures were created by Natalie (me) with ChemSketch and MarvinSketch, and they are free for you to use as long as you credit The Art of Paralytics as the source. I don't care if you have all the official citation format things, just as long as someone can tell where the picture came from. If you're doing a personal presentation like for a class and you want to use one of these pictures, feel free, and you don't need my permission. I'm not going to like go after you or sue you or something. In fact, I think it's awesome if someone uses my pictures.
Structures of stereoisomers were also determined by me, since there don't seem to be any other pictures online of mivacurium and doxacurium that show stereo bonds. The 3D structure of the acetylcholine receptor was obtained from the Protein Data Bank entry 2BG9 and the model was photographed in Chimera v1.8. All pictures are freely available for use under Creative Commons.
I do not claim any facts as my own - this page is not yet fully complete, and I am still gathering sources. Most of this information is stuff that I've read over many years that I don't remember the exact source for. However, I am working to compile as many sources as I can together that provide this information.
And yes, I've used the wikipedia pages for many of these, but I've double checked them to make sure they were correct. I am well aware that wikipedia can be wrong, so I have made sure that I was not.
Information on the origins of steroid structured NMBs:
Mckenzie AG. Prelude to pancuronium and vecuronium. Anaesthesia. 2000;55(6):551-6. - http://www.ncbi.nlm.nih.gov/pubmed/10866718
The acetylcholine receptor and binding site:
Goodsell D. Acetylcholine Receptor. Protein Data Bank. 2005 - http://www.rcsb.org/pdb/101/motm.do?momID=71