The 10 Atracurium Isomers
All 10 atracurium isomers are stereoisomers, meaning the atoms are arranged in the same order and have the same shape. The only thing that changes is the 3-d orientations of the bonds. This is just a list for reference.
The 10 atracurium isomers are:
The 10 atracurium isomers are:
- 1R-cis,1'R-cis
- 1R-cis, 1'R-trans ------ (same as 1R-trans, 1'R-cis)
- 1R-cis, 1'S-cis --------- (same as 1S-cis, 1'R-cis)
- 1R-cis, 1'S-trans ------ (same as 1S-trans, 1'R-cis)
- 1R-trans, 1'R-trans
- 1R-trans, 1'S-cis ------ (same as 1S-cis, 1'R-trans)
- 1R-trans, 1'S-trans --- (same as 1S-trans, 1'R-trans)
- 1S-cis, 1'S-cis
- 1S-cis, 1'S-trans ------ (same as 1S-trans, 1'S-cis)
- 1S-trans, 1'S-trans
How to figure out stereochemistry (1, 2, R, S, cis, and trans for atracurium isomers)
When I first got into neuromuscular blocking agents as an obsessive interest, I had never taken organic chemistry and had no idea what cisatracurium being 1R-cis, 1'R-cis meant. It was almost 2 years later, when I finally got to take an organic chemistry class, that I finally understood how the R, S, cis, and trans thing worked.
In order to understand how to name atracurium isomers, one must first analyze the name and break it down into parts. This will be done with cisatracurium's name as the primary example, but this can also be applied to any of the atracurium isomers.
Here is now a nice explanation for you. :)
You have the name:
1R-cis, 1'R-cis
Let's break it up into its parts.
1 R - cis, 1 ' R - cis
Now for an organic chemistry lesson.
In order to understand how to name atracurium isomers, one must first analyze the name and break it down into parts. This will be done with cisatracurium's name as the primary example, but this can also be applied to any of the atracurium isomers.
Here is now a nice explanation for you. :)
You have the name:
1R-cis, 1'R-cis
Let's break it up into its parts.
1 R - cis, 1 ' R - cis
Now for an organic chemistry lesson.
Step 1: Find the stereocenters. (the "1" or "2")

What is a stereocenter?
A stereocenter in organic chemistry is any atom in the molecule that has bonds to either 4 unique atoms, or 4 unique functional groups (parts of the molecule) if some bonds are to the same type of atom. This is done in such a way that the mirror image of the stereocenter atom and its surrounding atoms or structure cannot be superimposed onto the original. If that doesn't make sense, check out the picture on the right. Everything is easier to explain with pictures :)
These two imaginary molecules are mirror images. They consist of the same atoms, and the molecules are a reflection of each other. However, if you tried to rotate one of the molecules, no matter how much you twisted it around, you would not be able to get all the atoms of each color on one molecules to align perfectly with the colored atoms on another molecule. In other words, you can't superimpose one of the molecules on another. Therefore, even though they contain the same atoms, they are not the same molecules. A central atom that is bonded to 4 UNIQUE atoms or functional groups is considered a stereocenter. Because 4 unique bonds or functional groups are required, stereocenters will most likely be carbon (and sometimes nitrogen) atoms. In the picture above, the gray central atom is the stereocenter. An atom that is a stereocenter is called a chiral atom.
If you look near the bottom center of the picture at the structure made out of just lines (which is how they're usually drawn in organic chemistry), you will notice some of the bonds are in bold or are solid black triangles while others are dashed lines. Bold or triangle shaped bonds mean that in 3-d space, they are popping out towards your face. The bonds drawn as dashed lines are pointing away from you. This is very important to know. If it's hard to remember, just think bold is toward and dashed is away.
Another example is looking at your right hand vs. left hand. They're both hands, they have the same arrangement of fingers, but they are mirror images. You can't just superimpose one hand on another. With both palms facing the same way, try to line up the fingers on the right and left hands. Of course, it's not possible. The fingers on one hand don't match up with the other hand because even though they're reflections, they're still different! The same thing goes with some molecules.
Finding the stereocenters of atracurium
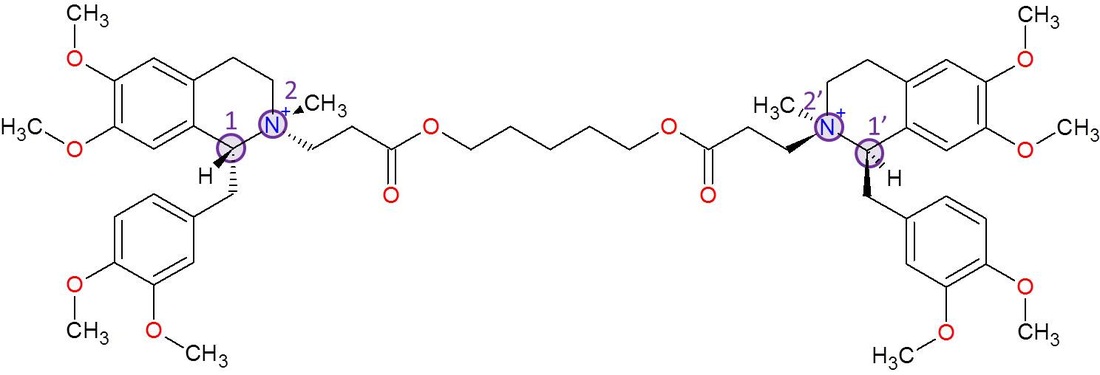
The atracurium molecule has 4 of these stereocenters, but they may be a bit hard to find. This is because the stereocenter atoms in atracurium have all 4 of their bonds to carbons or nitrogens rather than 4 unique atoms. However, the structure and shape of the parts of the molecule surrounding each bond coming off of the stereocenter are unique, so it still counts as a stereocenter.
Some atoms can be easily be eliminated when trying to find stereocenters. Hydrogens can only have 1 bond, and the oxygens only have 2 bonds, so they can't possibly be a stereocenter. This leaves the carbon and nitrogen atoms, which both form 4 bonds in this case. (Also, remember that with skeletal structures, when a carbon atom does not have 4 bonds to it, is is assumed that the remaining bonds are to a hydrogen. Every carbon atom in the structure has 4 bonds.)
It's pretty easy to eliminate carbons as well. For example, the ones on the aromatic rings have double bonds to the same carbon, meaning they don't have all 4 bonds to unique groups. The carbons on the upper part of non-aromatic rings only have 2 bonds to carbons shown - the other 2 bonds are to hydrogens, which means no 4 unique groups. If you eliminate all of the carbons with 2 or 3 bonds to hydrogen and all the carbons that have double bonds coming off of them, there are only 2 carbons left, one on each side of the molecule. Each of the bonds coming off of these carbons connect to unique parts of the molecule. These two carbons are stereocenters because their mirror images cannot be superimposed on them.
The other two stereocenters are actually the nitrogens. As with the vast majority of neuromuscular blocking agents, atracurium is a quaternary ammonium compound and has 4 bonds on its nitrogens. Not only does each nitrogen have 4 bonds, but those 4 bonds also are unique and go to unique parts of the molecule. Therefore, atracurium has 2 additional stereocenters with its nitrogen atoms, making 4 total.
Also a helpful hint if you're in any organic chemistry class - if you're asked to find stereocenters, look for any atom that has stereo bonds (bold or dashed line bonds). If all 4 bonds are unique, it's a stereocenter.
A stereocenter in organic chemistry is any atom in the molecule that has bonds to either 4 unique atoms, or 4 unique functional groups (parts of the molecule) if some bonds are to the same type of atom. This is done in such a way that the mirror image of the stereocenter atom and its surrounding atoms or structure cannot be superimposed onto the original. If that doesn't make sense, check out the picture on the right. Everything is easier to explain with pictures :)
These two imaginary molecules are mirror images. They consist of the same atoms, and the molecules are a reflection of each other. However, if you tried to rotate one of the molecules, no matter how much you twisted it around, you would not be able to get all the atoms of each color on one molecules to align perfectly with the colored atoms on another molecule. In other words, you can't superimpose one of the molecules on another. Therefore, even though they contain the same atoms, they are not the same molecules. A central atom that is bonded to 4 UNIQUE atoms or functional groups is considered a stereocenter. Because 4 unique bonds or functional groups are required, stereocenters will most likely be carbon (and sometimes nitrogen) atoms. In the picture above, the gray central atom is the stereocenter. An atom that is a stereocenter is called a chiral atom.
If you look near the bottom center of the picture at the structure made out of just lines (which is how they're usually drawn in organic chemistry), you will notice some of the bonds are in bold or are solid black triangles while others are dashed lines. Bold or triangle shaped bonds mean that in 3-d space, they are popping out towards your face. The bonds drawn as dashed lines are pointing away from you. This is very important to know. If it's hard to remember, just think bold is toward and dashed is away.
Another example is looking at your right hand vs. left hand. They're both hands, they have the same arrangement of fingers, but they are mirror images. You can't just superimpose one hand on another. With both palms facing the same way, try to line up the fingers on the right and left hands. Of course, it's not possible. The fingers on one hand don't match up with the other hand because even though they're reflections, they're still different! The same thing goes with some molecules.
Finding the stereocenters of atracurium
The atracurium molecule has 4 of these stereocenters, but they may be a bit hard to find. This is because the stereocenter atoms in atracurium have all 4 of their bonds to carbons or nitrogens rather than 4 unique atoms. However, the structure and shape of the parts of the molecule surrounding each bond coming off of the stereocenter are unique, so it still counts as a stereocenter.
Some atoms can be easily be eliminated when trying to find stereocenters. Hydrogens can only have 1 bond, and the oxygens only have 2 bonds, so they can't possibly be a stereocenter. This leaves the carbon and nitrogen atoms, which both form 4 bonds in this case. (Also, remember that with skeletal structures, when a carbon atom does not have 4 bonds to it, is is assumed that the remaining bonds are to a hydrogen. Every carbon atom in the structure has 4 bonds.)
It's pretty easy to eliminate carbons as well. For example, the ones on the aromatic rings have double bonds to the same carbon, meaning they don't have all 4 bonds to unique groups. The carbons on the upper part of non-aromatic rings only have 2 bonds to carbons shown - the other 2 bonds are to hydrogens, which means no 4 unique groups. If you eliminate all of the carbons with 2 or 3 bonds to hydrogen and all the carbons that have double bonds coming off of them, there are only 2 carbons left, one on each side of the molecule. Each of the bonds coming off of these carbons connect to unique parts of the molecule. These two carbons are stereocenters because their mirror images cannot be superimposed on them.
The other two stereocenters are actually the nitrogens. As with the vast majority of neuromuscular blocking agents, atracurium is a quaternary ammonium compound and has 4 bonds on its nitrogens. Not only does each nitrogen have 4 bonds, but those 4 bonds also are unique and go to unique parts of the molecule. Therefore, atracurium has 2 additional stereocenters with its nitrogen atoms, making 4 total.
Also a helpful hint if you're in any organic chemistry class - if you're asked to find stereocenters, look for any atom that has stereo bonds (bold or dashed line bonds). If all 4 bonds are unique, it's a stereocenter.
In this picture, I added the hydrogens to the chiral carbons just so you can see all 4 bonds. The stereocenters are also numbered. When numbering the carbons in atracurium, the one with the "1" label is actually carbon number 1, and nitrogen, being right next to it, gets the 2 label. The reason there are two 1's and two 2's is because if you look at the shape of atracurium, it is also symmetrical. You could flip it around, and it would still have the same general structure and order of bonds. There is no specific reason to assign the carbon on the left a higher or lower number than the one on the right since the two are basically the same. So one of the carbons simply gets a ' after its number so that you know that the 1 and 1' carbons are still two separate carbons within the entire molecule. The same is true with the nitrogen labeled 2 and 2'. The next section will show more clearly how they are stereocenters if you're still not sure.
Step 2: Determine R or S
Since molecules with stereocenters often cannot be superimposed on each other as you know from the first part, they are considered to be different molecules. These different molecules are known as stereoisomers. This is important because with many molecules, not just atracurium, different stereoisomers have different pharmacological effects. But how do you properly differentiate one stereoisomer from another?
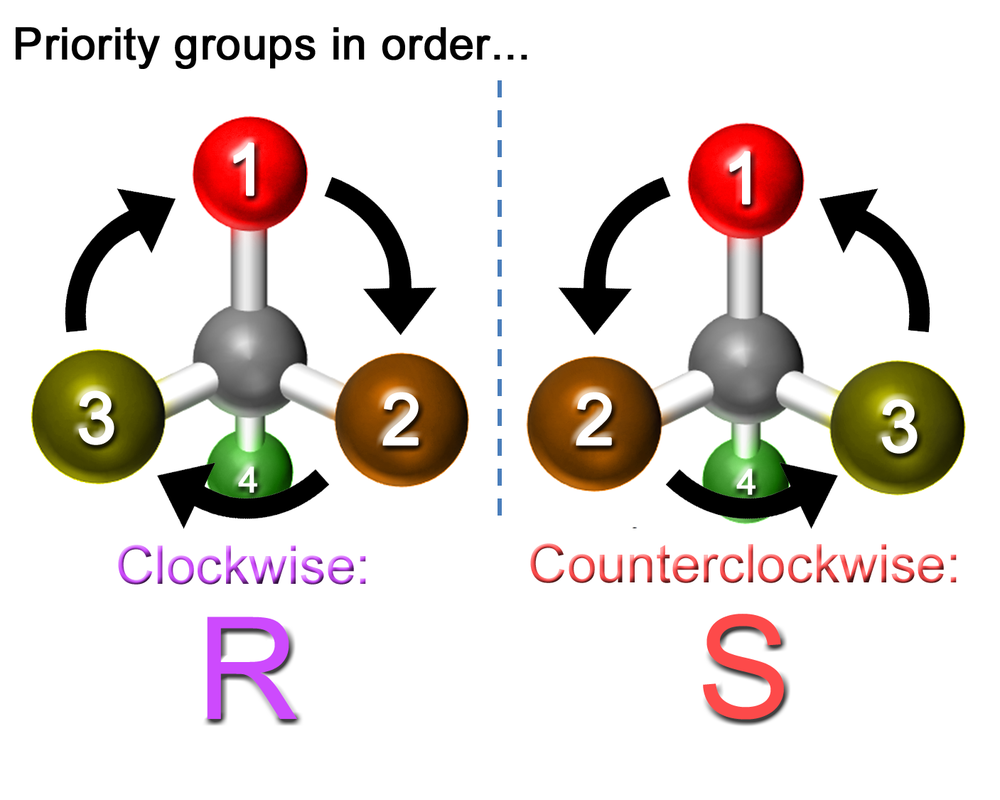
Since stereoisomers exist as mirror images of each other, there are 2 possibilities for each orientation of a stereocenter - either one orientation or its mirror image. Organic chemists use the terms R and S to tell which orientation is which. One of the mirror images will be R, and the other will be S.
If you're going to have a label to differentiate stereocenter orientations, you should be able to tell which orientation is which, whether it is R or S. Luckily, it's relatively easy to learn how to do it. What's more difficult is actually putting it into practice and labeling the stereocenters yourself because it requires some spatial skills and the ability to rotate and visualize 3D objects in your mind. Alternatively, you can do it the "easy way," which doesn't require spatial skills, and that way will also be explained.
Getting your priorities straight
After you've found a stereocenter, the first step to determining R or S is to give each bond/atom coming off the stereocenter a priority number. Since there are 4 unique bonds coming off the stereocenter, each one will be given a priority from 1 to 4. It's really easy, and all you'll need is either your knowledge of chemistry or a periodic table.
What if there's a tie?
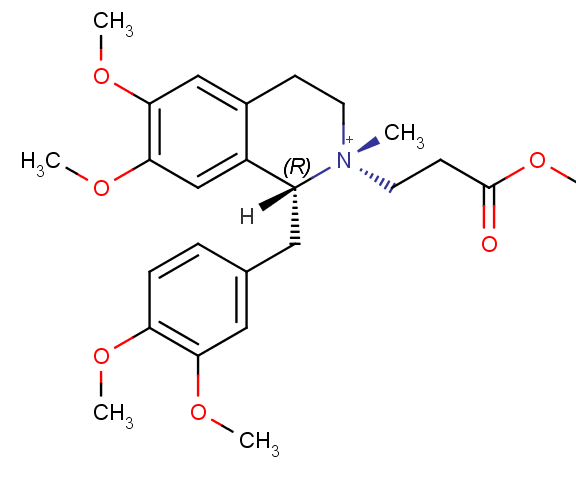
If there's a tie, then you go one more atom out from there in the direction that would result in the highest priority for that next atom out. See which atoms out of the remaining ones have the highest priority. It's like when there's a tie playing the card game War. You just keep doing tiebreakers over and over again until somebody wins. Like always, a picture's worth a thousand words, especially in organic chemistry. Here is cisatracurium and how to determine whether the "1" stereocenter (the carbon on the left half of the molecule) is R or S.
Below is the left half of cisatracurium. The circled carbon is the stereocenter. Click the arrows on the picture to progress to the next step in figuring out the stereochemistry.
Since stereoisomers exist as mirror images of each other, there are 2 possibilities for each orientation of a stereocenter - either one orientation or its mirror image. Organic chemists use the terms R and S to tell which orientation is which. One of the mirror images will be R, and the other will be S.
If you're going to have a label to differentiate stereocenter orientations, you should be able to tell which orientation is which, whether it is R or S. Luckily, it's relatively easy to learn how to do it. What's more difficult is actually putting it into practice and labeling the stereocenters yourself because it requires some spatial skills and the ability to rotate and visualize 3D objects in your mind. Alternatively, you can do it the "easy way," which doesn't require spatial skills, and that way will also be explained.
Getting your priorities straight
After you've found a stereocenter, the first step to determining R or S is to give each bond/atom coming off the stereocenter a priority number. Since there are 4 unique bonds coming off the stereocenter, each one will be given a priority from 1 to 4. It's really easy, and all you'll need is either your knowledge of chemistry or a periodic table.
- Step 1: Look at all the bonds coming off of your stereocenter and see which atom bonded to it has the highest atomic number. That one is the highest priority, or priority 1.
- Step 2: Look at the remaining atoms and do the same. Label the next highest priority atom 2, and then 3, and the lowest 4.
What if there's a tie?
If there's a tie, then you go one more atom out from there in the direction that would result in the highest priority for that next atom out. See which atoms out of the remaining ones have the highest priority. It's like when there's a tie playing the card game War. You just keep doing tiebreakers over and over again until somebody wins. Like always, a picture's worth a thousand words, especially in organic chemistry. Here is cisatracurium and how to determine whether the "1" stereocenter (the carbon on the left half of the molecule) is R or S.
Below is the left half of cisatracurium. The circled carbon is the stereocenter. Click the arrows on the picture to progress to the next step in figuring out the stereochemistry.
Rotating the Bonds
This next step is where your spatial skills, if you have them, will come in handy. It's also important to remember that bonds drawn with bold lines or triangles are coming toward you and bonds drawn with dashed lines are going away from you.
First, you want to rearrange the bonds in the molecule by rotating the stereocenter so that the group with the LOWEST priority is in the back going away from you, meaning it is coming from a dashed line. In this example with atracurium, you want to mentally rotate the molecule and position the hydrogen in the back at the end of a dashed line. Right now, since the bond to hydrogen is bolded, the hydrogen is facing toward you. You want to make it face away from you. If you have the spatial skills to remember the orientations of all 4 bonds as you rotate the molecule in your mind while remembering the priority of each, go ahead and do that. If you don't have spatial skills or you don't want to waste mental energy, there's a better way.
First, draw only the stereocenter and the four bonds coming off of the stereocenter. This is simply because doing multiple drawings of trying to rotate a large molecule is a huge pain. Doing just the stereocenter and its bonds is much easier. Be sure to specify which bond is which priority if there were any ties in the previous step.
Now pick one of the bonds that isn't a stereo bond (pick one that's just a straight line), and make sure that the one you pick also isn't the bond to the atom with 4th priority. This bond will be held constant, and you will be rotating the molecule around it to position the lowest priority atom in the back. In this case, either the first or 2nd priority bond can be held constant. For this example, I'll be keeping the nitrogen constant and rotating the bonds around it. Then, move each number down or over one position so that the lowest priority atom is on the dashed bonds. There will be one atom that has no new position to move to - move that one all the way back around to the beginning. It's easier to explain with pictures.
First, after choosing the atom to hold constant, draw the stereocenter by itself with just the first bond and the numbers coming off of it. Then, shift the other three numbered bonds one position so that the 4th priority bond is coming from the dashed line.
This next step is where your spatial skills, if you have them, will come in handy. It's also important to remember that bonds drawn with bold lines or triangles are coming toward you and bonds drawn with dashed lines are going away from you.
First, you want to rearrange the bonds in the molecule by rotating the stereocenter so that the group with the LOWEST priority is in the back going away from you, meaning it is coming from a dashed line. In this example with atracurium, you want to mentally rotate the molecule and position the hydrogen in the back at the end of a dashed line. Right now, since the bond to hydrogen is bolded, the hydrogen is facing toward you. You want to make it face away from you. If you have the spatial skills to remember the orientations of all 4 bonds as you rotate the molecule in your mind while remembering the priority of each, go ahead and do that. If you don't have spatial skills or you don't want to waste mental energy, there's a better way.
First, draw only the stereocenter and the four bonds coming off of the stereocenter. This is simply because doing multiple drawings of trying to rotate a large molecule is a huge pain. Doing just the stereocenter and its bonds is much easier. Be sure to specify which bond is which priority if there were any ties in the previous step.
Now pick one of the bonds that isn't a stereo bond (pick one that's just a straight line), and make sure that the one you pick also isn't the bond to the atom with 4th priority. This bond will be held constant, and you will be rotating the molecule around it to position the lowest priority atom in the back. In this case, either the first or 2nd priority bond can be held constant. For this example, I'll be keeping the nitrogen constant and rotating the bonds around it. Then, move each number down or over one position so that the lowest priority atom is on the dashed bonds. There will be one atom that has no new position to move to - move that one all the way back around to the beginning. It's easier to explain with pictures.
First, after choosing the atom to hold constant, draw the stereocenter by itself with just the first bond and the numbers coming off of it. Then, shift the other three numbered bonds one position so that the 4th priority bond is coming from the dashed line.
|
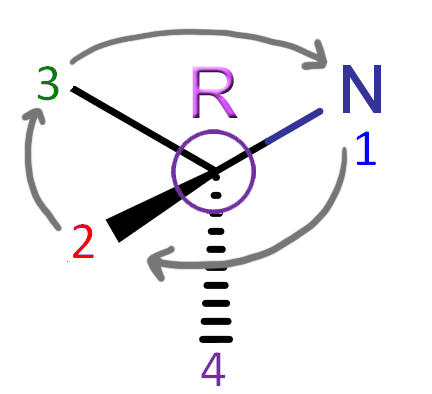
Determining R or S After you have the 4th priority bond facing away from you, determining if a stereocenter is R or S is really easy. Now, look at the first 3 bonds, and counting from 1 to 3, see if they're arranged clockwise or counterclockwise in front of the 4th priority bond. If the bonds going 1 to 3 are arranged clockwise, the stereocenter is R. If they are counterclockwise, the stereocenter is S. Now, we can apply this to the stereocenter we found in atracurium! |
Step 3: Determine Cis or Trans
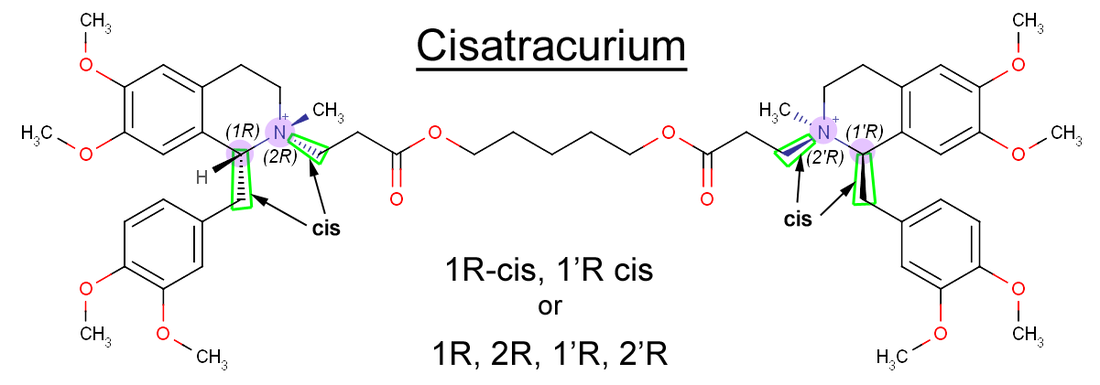
Whether two bonds are cis and trans to each other can be determined easily just by looking at the molecule. Two bonds or functional groups that are cis to each other will be facing the same way, either both toward you or both away from you. Two bonds that are trans, on the other hand, face opposite directions. In atracurium, the two bonds that are compared when determining cis or trans are two of the bonds coming off of the stereocenters. More specifically, it's these two bonds shown below in green:

Because these two bonds are both dashed, meaning they're both facing in the same direction, they are considered to be cis. The carbon that we already labeled as R is carbon 1. Therefore, the first part of the name of the isomer is 1R-cis.
If you take the right half of the molecule and figure out its stereochemistry, you'll find that the carbon stereocenter on the other side is R, and the bonds are cis as well. Since both halves of the molecule are symmetrical and equivalent, the carbon on the right half is simply called 1' instead of 1.
Therefore, the full name of the isomer is 1R-cis, 1'R-cis.
In addition, the two quaternary ammoniums themselves are also stereocenters, and they are also R. They are labeled with the number 2. Therefore, some people name the cisatracurium isomer based on the chirality of all 4 stereocenters instead of the cis/trans orientation. In such a case, the isomer can also be called 1R, 2R, 1'R, 2'R. In atracurium (but not in all molecules), when both stereocenters on one side are the same (R or S), the bonds on that side will also be cis, so the second name also implies that the two bonds shown in green are cis to each other.
Below is cisatracurium with all four stereocenters labeled. The carbons and nitrogens are also numbered. In cisatracurium, all stereocenters are R.
If you take the right half of the molecule and figure out its stereochemistry, you'll find that the carbon stereocenter on the other side is R, and the bonds are cis as well. Since both halves of the molecule are symmetrical and equivalent, the carbon on the right half is simply called 1' instead of 1.
Therefore, the full name of the isomer is 1R-cis, 1'R-cis.
In addition, the two quaternary ammoniums themselves are also stereocenters, and they are also R. They are labeled with the number 2. Therefore, some people name the cisatracurium isomer based on the chirality of all 4 stereocenters instead of the cis/trans orientation. In such a case, the isomer can also be called 1R, 2R, 1'R, 2'R. In atracurium (but not in all molecules), when both stereocenters on one side are the same (R or S), the bonds on that side will also be cis, so the second name also implies that the two bonds shown in green are cis to each other.
Below is cisatracurium with all four stereocenters labeled. The carbons and nitrogens are also numbered. In cisatracurium, all stereocenters are R.
Why are there 10 isomers?
Each stereocenter in a molecule can be oriented one of two ways, R or S. Therefore, to figure out the number of stereoisomers, all you have to do is take 2 raised to the power of the number of stereocenters. Atracurium has 4 stereocenters. 2 raised to the 4th power is 16, so there are 16 possible isomers of atracurium.
"16? I thought you said there were only 10 isomers!"
Yes, there are only 10 actual isomers of atracurium. This is because 6 of the 16 theoretical isomers are just repeats of other isomers.
Because atracurium has bilateral symmetry, the molecule can be rotated around its line of symmetry until it faces backwards while still maintaining the same exact overall shape as if it had never been rotated at all. There are 6 isomers, which, when rotated to face backwards, are found to be the same exact molecule as another hypothetical isomer. Therefore, 6 of the 16 possible isomers are just repeats of other isomers, leaving just 10 unique isomers.
As an example, take the binary numbers from 0000 to 1111. Each digit represents one of the four stereocenters, which can each potentially be oriented in one of two ways, represented by 0 or 1, and there are 16 possible combinations of 0 and 1.
The numbers 0000 and 1111, as well as 0110 and 1001, are palindromes. If they are flipped around, they are still the same number. However, something like 1010 can be flipped around to make 0101. Since molecules can face any direction while in 3D space, if 1010 and 0101 were two molecules, they would be considered to be the same thing, just rotated in a different direction. 1100 would be the same molecule as 0011, just flipped around.
So you start listing all the different atracurium isomers. You start listing all the binary combinations... 0000, 0001, 0010, 0011, 0100...
Oh, wait: 0100 is the same thing as 0010, just flipped around. This means 0100 doesn't count as a separate isomer. As you continue counting, you notice a total of 6 numbers that are the same as 6 previous numbers when flipped around.
0100 = 0010
0101 = 1010
0111 = 1110
0001 = 1000
0011 = 1100
1011 = 1101
Since each member of the pair is the same as another when flipped around, each pair only counts as 1 isomer. With those six pairs of two, as well as the four palindromes, 0000, 1111, 0110, and 1001, there are only 10 unique isomers, even though there are 16 theoretical combinations.
Congratulations! You now know more about atracurium and its isomers than you'll ever need to!
Sources:
Making sure of which stereocenter is 1 or 2 http://engineering.verticalnews.com/articles/9656001.html
Cis vs. Trans atracurium structure pictures http://www.tlcpharmachem.com/tlc_item.php?upc=A-756&li=&sub=
Organic chemistry information: Smith, J. (2011). Organic chemistry. New York, NY: McGraw-Hill.
All pictures on this page were created by Natalie and are available for use under Creative Commons. All molecule drawings were created using MarvinSketch and Adobe Photoshop Elements.
"16? I thought you said there were only 10 isomers!"
Yes, there are only 10 actual isomers of atracurium. This is because 6 of the 16 theoretical isomers are just repeats of other isomers.
Because atracurium has bilateral symmetry, the molecule can be rotated around its line of symmetry until it faces backwards while still maintaining the same exact overall shape as if it had never been rotated at all. There are 6 isomers, which, when rotated to face backwards, are found to be the same exact molecule as another hypothetical isomer. Therefore, 6 of the 16 possible isomers are just repeats of other isomers, leaving just 10 unique isomers.
As an example, take the binary numbers from 0000 to 1111. Each digit represents one of the four stereocenters, which can each potentially be oriented in one of two ways, represented by 0 or 1, and there are 16 possible combinations of 0 and 1.
The numbers 0000 and 1111, as well as 0110 and 1001, are palindromes. If they are flipped around, they are still the same number. However, something like 1010 can be flipped around to make 0101. Since molecules can face any direction while in 3D space, if 1010 and 0101 were two molecules, they would be considered to be the same thing, just rotated in a different direction. 1100 would be the same molecule as 0011, just flipped around.
So you start listing all the different atracurium isomers. You start listing all the binary combinations... 0000, 0001, 0010, 0011, 0100...
Oh, wait: 0100 is the same thing as 0010, just flipped around. This means 0100 doesn't count as a separate isomer. As you continue counting, you notice a total of 6 numbers that are the same as 6 previous numbers when flipped around.
0100 = 0010
0101 = 1010
0111 = 1110
0001 = 1000
0011 = 1100
1011 = 1101
Since each member of the pair is the same as another when flipped around, each pair only counts as 1 isomer. With those six pairs of two, as well as the four palindromes, 0000, 1111, 0110, and 1001, there are only 10 unique isomers, even though there are 16 theoretical combinations.
Congratulations! You now know more about atracurium and its isomers than you'll ever need to!
Sources:
Making sure of which stereocenter is 1 or 2 http://engineering.verticalnews.com/articles/9656001.html
Cis vs. Trans atracurium structure pictures http://www.tlcpharmachem.com/tlc_item.php?upc=A-756&li=&sub=
Organic chemistry information: Smith, J. (2011). Organic chemistry. New York, NY: McGraw-Hill.
All pictures on this page were created by Natalie and are available for use under Creative Commons. All molecule drawings were created using MarvinSketch and Adobe Photoshop Elements.